MR Spectroscopy of the Brain in Chornomorsk – Advanced Neuro MRI Metabolic Analysis on Philips Achieva 1.5 Tesla
Magnetic Resonance Spectroscopy (MR Spectroscopy, MRS, Proton Spectroscopy, 1H-MRS) is an advanced neuroradiology MRI technique that evaluates the biochemical and metabolic composition of brain tissue. Unlike conventional MRI, which primarily demonstrates anatomical structures and morphological abnormalities, MR spectroscopy analyzes tissue metabolism and metabolite concentration patterns within selected brain regions.
At MEDESSA diagnostic center in Chornomorsk, brain MR spectroscopy is performed on the Philips Achieva 1.5 Tesla MRI system as part of comprehensive neuro MRI protocols. Modern 1.5 Tesla MR spectroscopy remains clinically valuable for evaluation of brain tumors, epilepsy, demyelinating disease, inflammatory disorders, postoperative changes, neurodegeneration, and metabolic brain abnormalities when combined with high-quality structural MRI sequences.
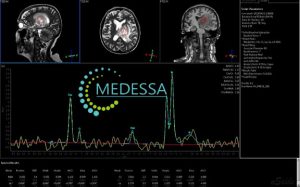
MR spectroscopy is considered one of the most advanced functional MRI methods in modern neuroradiology because it allows indirect non-invasive assessment of neuronal integrity, membrane turnover, anaerobic metabolism, cellular proliferation, necrosis, gliosis, inflammation, and tumor aggressiveness.
What Is Brain MR Spectroscopy
MR spectroscopy is a specialized MRI technique that measures chemical metabolites within brain tissue. Instead of generating only anatomical images, spectroscopy produces a metabolic spectrum containing peaks corresponding to specific biochemical compounds.
The examination evaluates intracellular metabolic activity and tissue biochemistry using proton resonance frequencies. Changes in metabolite concentrations may occur before visible structural abnormalities become apparent on conventional MRI.
Brain MR spectroscopy may help differentiate tumor recurrence from radiation necrosis, distinguish neoplastic from non-neoplastic lesions, evaluate epileptogenic cortex, assess demyelinating disease activity, and improve characterization of metabolic or neurodegenerative disorders.
What MR Spectroscopy Shows
Brain MR spectroscopy provides metabolic information about brain tissue composition and cellular activity.
Depending on the pathology, spectroscopy may demonstrate:
- Neuronal loss or dysfunction;
- Increased membrane turnover;
- Tumor proliferation;
- Necrosis;
- Hypoxic injury;
- Anaerobic metabolism;
- Inflammatory changes;
- Demyelination;
- Gliosis;
- Altered cellular energetics;
- Metabolic asymmetry in epilepsy;
- Treatment response after surgery or radiotherapy.
How MR Spectroscopy Differs From Standard Brain MRI
Conventional MRI primarily evaluates anatomical and structural abnormalities, while MR spectroscopy evaluates tissue metabolism and biochemical composition.
| Standard MRI | MR Spectroscopy |
|---|---|
| Evaluates anatomy | Evaluates metabolism |
| Shows morphology | Shows biochemical activity |
| Detects edema and lesions | Analyzes metabolite changes |
| Visualizes structural abnormalities | Characterizes tissue chemistry |
| Uses image interpretation | Uses spectral analysis |
| Essential for localization | Useful for lesion characterization |
MR spectroscopy does not replace standard brain MRI. Spectroscopy is an advanced complementary method that should be interpreted together with conventional MRI sequences.
Why MR Spectroscopy Must Be Performed Together With Standard Brain MRI
MR spectroscopy alone has limited diagnostic value without complete structural MRI evaluation. Conventional MRI sequences remain essential for lesion localization, anatomical assessment, edema evaluation, hemorrhage detection, enhancement analysis, vascular assessment, and tissue characterization.
In modern neuroradiology practice, standard MRI is performed first to evaluate morphology and anatomical abnormalities. Spectroscopy is then used as an advanced metabolic analysis tool.
Without structural MRI sequences:
- Voxel placement may be inaccurate;
- Metabolic abnormalities may be misinterpreted;
- Tumor heterogeneity may be underestimated;
- Necrotic regions may not be distinguished correctly;
- Hemorrhage and calcification may mimic metabolic changes;
- Artifacts may significantly reduce interpretation accuracy.
For this reason, brain MR spectroscopy should always be integrated into a comprehensive neuro MRI protocol.
Why Spectroscopy Should Be Combined With Advanced Neuro MRI Sequences
T1-Weighted Imaging
T1 sequences provide anatomical localization and tissue morphology assessment. They are essential for accurate voxel positioning and lesion boundary evaluation.
T2-Weighted Imaging
T2 imaging helps identify edema, cystic degeneration, gliosis, inflammatory changes, and lesion extent.
FLAIR
FLAIR sequences are critically important for detecting white matter lesions, demyelination, cortical abnormalities, and infiltrative tumor margins.
DWI and ADC
DWI and ADC assess tissue diffusion characteristics and cellularity. Combined with spectroscopy, they improve differentiation between abscess, lymphoma, high-grade tumors, ischemia, and treatment-related changes.
SWI
SWI detects hemorrhage, calcification, microbleeds, and vascular abnormalities that may influence spectroscopic interpretation.
T1 With Contrast Enhancement
Contrast-enhanced MRI improves lesion targeting and helps identify active tumor regions with increased vascularity and blood-brain barrier disruption.
Perfusion MRI
Perfusion imaging evaluates vascularity and cerebral blood volume. Combined perfusion and spectroscopy improve grading of gliomas and differentiation between recurrence and radionecrosis.
Tractography
Diffusion tensor imaging and tractography help assess white matter tract displacement or infiltration in brain tumors and preoperative planning.
MR Angiography
MR angiography evaluates vascular anatomy, aneurysms, arterial stenosis, vascular malformations, and tumor vascular supply.
Which MRI Sequences Are Used Together With Brain MR Spectroscopy
Modern spectroscopy protocols are integrated into multiparametric neuro MRI examinations.
T1
Provides anatomical reference and lesion localization.
T2
Evaluates edema, cystic changes, and lesion extent.
FLAIR
Improves detection of infiltrative white matter abnormalities and demyelination.
DWI
Assesses restricted diffusion and acute cellular injury.
ADC
Helps differentiate true diffusion restriction from T2 shine-through effects.
SWI
Detects hemorrhage and susceptibility-related abnormalities.
T1+C
Evaluates enhancement patterns and active tumor regions.
Perfusion MRI
Measures cerebral blood volume and vascularity.
TOF MRA
Evaluates intracranial vascular structures and vascular pathology.
Which Diseases MR Spectroscopy Helps Diagnose
- Glioma;
- Glioblastoma;
- Brain metastases;
- Primary CNS lymphoma;
- Radionecrosis;
- Temporal lobe epilepsy;
- Multiple sclerosis;
- Encephalitis;
- Brain abscess;
- Dementia;
- Neurodegenerative disorders;
- Hypoxic brain injury;
- Toxic encephalopathy;
- Metabolic brain disease;
- Mitochondrial disorders;
- Post-treatment tumor recurrence.
MR Spectroscopy in Brain Tumors
Brain tumors are among the most important indications for MR spectroscopy.
Spectroscopy may help evaluate:
- Tumor aggressiveness;
- Cellular proliferation;
- Necrosis;
- Tumor infiltration;
- Peritumoral edema;
- Residual tumor after surgery;
- Treatment response;
- Radiation necrosis versus recurrence.
High-grade gliomas often demonstrate:
- Elevated choline;
- Reduced NAA;
- Increased Cho/NAA ratio;
- Lactate peaks;
- Lipid peaks associated with necrosis.
MR Spectroscopy in Epilepsy
MR spectroscopy may help identify metabolic abnormalities within epileptogenic cortex even when structural MRI findings are subtle.
Temporal lobe epilepsy may demonstrate:
- Reduced NAA;
- Neuronal dysfunction;
- Metabolic asymmetry between hemispheres;
- Hippocampal metabolic abnormalities.
Spectroscopy may improve preoperative epilepsy evaluation together with dedicated epilepsy MRI protocols.
MR Spectroscopy in Demyelinating Disease
In multiple sclerosis and other demyelinating disorders, spectroscopy may reveal:
- Reduced neuronal integrity;
- Gliosis;
- Inflammatory metabolic changes;
- Axonal injury;
- Altered myo-inositol levels.
Spectroscopy findings may complement FLAIR and contrast-enhanced MRI assessment of disease activity.
MR Spectroscopy in Neurodegenerative Disorders
Neurodegenerative disease may demonstrate altered neuronal metabolism before advanced structural atrophy becomes visible.
Potential applications include:
- Alzheimer disease;
- Frontotemporal dementia;
- Parkinsonian syndromes;
- Mitochondrial disorders;
- Hypoxic brain injury.
MR Spectroscopy After Surgery and Radiation Therapy
Post-treatment neuroimaging interpretation is often challenging. MR spectroscopy may help differentiate:
- Tumor recurrence;
- Radiation necrosis;
- Postoperative gliosis;
- Treatment-related inflammation;
- Residual viable tumor tissue.
Combined perfusion MRI and spectroscopy significantly improve diagnostic confidence in neuro-oncology follow-up.
Which Metabolites Are Evaluated During MR Spectroscopy
- N-acetylaspartate (NAA);
- Choline (Cho);
- Creatine (Cr);
- Lactate;
- Lipids;
- Myo-inositol;
- Glutamate and glutamine;
- Alanine in selected lesions.
What NAA, Choline, Creatine, Lactate, Lipids, and Myo-Inositol Peaks Mean
NAA (N-Acetylaspartate)
NAA is considered a neuronal marker. Reduced NAA may indicate neuronal loss or dysfunction.
Choline
Choline reflects membrane turnover and cellular proliferation. Elevated choline is commonly associated with tumors and active demyelination.
Creatine
Creatine is related to cellular energy metabolism and often serves as a metabolic reference marker.
Lactate
Lactate may indicate anaerobic metabolism, hypoxia, ischemia, abscess, or aggressive tumors.
Lipids
Lipid peaks are often associated with necrosis, aggressive neoplasms, and tissue breakdown.
Myo-Inositol
Myo-inositol may increase in gliosis, astrocytosis, and some neurodegenerative disorders.
What the Cho/NAA Ratio Means
The choline-to-NAA ratio is one of the most important spectroscopy parameters in neuro-oncology.
An elevated Cho/NAA ratio may suggest:
- Increased tumor cellularity;
- Higher tumor grade;
- Active tumor proliferation;
- Reduced neuronal integrity.
Cho/NAA analysis is commonly used in glioma characterization and treatment monitoring.
Types of Brain MR Spectroscopy
Single Voxel Spectroscopy (SVS)
SVS evaluates metabolism within a single selected region of interest. It is commonly used for focal lesion characterization.
Multi Voxel Spectroscopy / Chemical Shift Imaging (CSI)
Multi-voxel spectroscopy evaluates multiple regions simultaneously and produces metabolic maps across larger tissue volumes.
CSI is especially useful for:
- Tumor heterogeneity assessment;
- Infiltrative glioma evaluation;
- Metabolic mapping;
- Preoperative planning.
How MR Spectroscopy Is Performed
MR spectroscopy is performed as part of a comprehensive brain MRI examination.
The process usually includes:
- Structural MRI acquisition;
- Lesion localization;
- Voxel positioning;
- Spectral acquisition;
- Metabolite analysis;
- Correlation with anatomical MRI findings.
Which Technical Methods Are Used in MR Spectroscopy
PRESS Technique
PRESS (Point Resolved Spectroscopy) is one of the most commonly used spectroscopy acquisition techniques because of its high signal efficiency.
STEAM Technique
STEAM allows shorter echo times and may improve evaluation of selected metabolites.
Short TE
Short echo time protocols detect a broader range of metabolites but may produce more complex spectra.
Long TE
Long TE protocols simplify spectral interpretation and improve lactate detection.
Suppression Techniques
Water and fat suppression methods improve spectral quality and reduce artifacts.
Voxel Positioning
Accurate voxel placement is critically important to avoid contamination from necrosis, hemorrhage, CSF, bone, or adjacent tissues.
Spectral Mapping
Metabolic mapping allows evaluation of heterogeneous lesions and infiltrative tumor margins.
Why MR Spectroscopy Is Especially Informative at 3.0 Tesla
Higher magnetic field strength improves spectroscopy performance because of increased signal-to-noise ratio and better spectral separation.
Advantages of 3.0 Tesla spectroscopy may include:
- Higher signal-to-noise ratio;
- Improved metabolite resolution;
- Better spectral separation;
- More accurate small lesion evaluation;
- Improved tumor characterization;
- Enhanced detection of subtle metabolic abnormalities.
However, modern optimized 1.5 Tesla MR spectroscopy on high-quality systems such as Philips Achieva remains clinically useful and widely applied in routine neuroradiology practice worldwide.
When Brain MR Spectroscopy Is Recommended
- Glioma evaluation;
- Glioblastoma characterization;
- Brain metastases;
- Primary CNS lymphoma;
- Differentiation of radionecrosis and recurrence;
- Epilepsy evaluation;
- Multiple sclerosis;
- Encephalitis;
- Brain abscess;
- Dementia assessment;
- Neurodegenerative disease;
- Hypoxic injury;
- Toxic encephalopathy;
- Metabolic brain disorders;
- Post-treatment monitoring.
Related MRI Examinations
Frequently Asked Questions About Brain MR Spectroscopy
What is brain MR spectroscopy?
Brain MR spectroscopy is an advanced MRI technique that evaluates tissue metabolism and biochemical composition rather than only anatomical structures.
Does MR spectroscopy replace standard brain MRI?
No. MR spectroscopy is an advanced complementary technique and should always be interpreted together with conventional MRI sequences.
What does MR spectroscopy detect?
MR spectroscopy evaluates metabolites associated with neuronal integrity, tumor proliferation, necrosis, inflammation, demyelination, and cellular metabolism.
Can spectroscopy diagnose brain tumors?
Spectroscopy may improve tumor characterization and help differentiate tumor types, grades, recurrence, and treatment-related changes.
What is the Cho/NAA ratio?
The Cho/NAA ratio reflects the relationship between cellular proliferation and neuronal integrity and is widely used in neuro-oncology assessment.
Can MR spectroscopy detect epilepsy?
Spectroscopy may demonstrate metabolic abnormalities within epileptogenic cortex, particularly in temporal lobe epilepsy.
What metabolites are evaluated during spectroscopy?
Common metabolites include NAA, choline, creatine, lactate, lipids, and myo-inositol.
Is MR spectroscopy useful in multiple sclerosis?
Yes. Spectroscopy may reveal metabolic evidence of demyelination, axonal injury, gliosis, and inflammatory changes.
Why is spectroscopy useful after radiation therapy?
Spectroscopy may help differentiate recurrent tumor from radionecrosis and treatment-related tissue changes.
Is MR spectroscopy performed with contrast?
Spectroscopy itself does not require contrast, but it is often combined with contrast-enhanced MRI for comprehensive lesion characterization.
What is the difference between SVS and CSI spectroscopy?
SVS analyzes a single selected region, while CSI evaluates multiple regions and produces metabolic maps.
Why is spectroscopy more informative at 3 Tesla?
Higher magnetic field strength improves signal-to-noise ratio, spectral separation, and metabolite resolution.
Can spectroscopy detect radiation necrosis?
Combined spectroscopy and perfusion MRI may improve differentiation between radionecrosis and recurrent tumor.
Can spectroscopy diagnose dementia?
Spectroscopy may demonstrate metabolic abnormalities associated with neurodegenerative disorders, although interpretation must always be correlated with clinical and structural imaging findings.
How long does brain MR spectroscopy take?
The spectroscopy component usually adds additional acquisition time to the standard MRI examination depending on protocol complexity.
